Cancer isn’t just a disease of uncontrolled cell growth—it’s a complex metabolic puzzle. Every tumor has unique energy demands, adapting its metabolism to survive, grow, and thrive in diverse environments. Understanding cancer metabolism gives you insight into why certain tumors behave aggressively and how therapies target these metabolic pathways.
In this comprehensive guide, you’ll discover what fuels different tumor types, why metabolic diversity matters, and how researchers are exploring innovative strategies to disrupt cancer’s energy supply. By the end, you’ll have a clearer understanding of the biological engines behind cancer growth and what that means for treatment strategies.
Meta description (150–160 characters): Explore cancer metabolism, learn what fuels various tumor types, and discover how metabolic differences shape tumor growth and treatment strategies.
The Basics of Cancer Metabolism
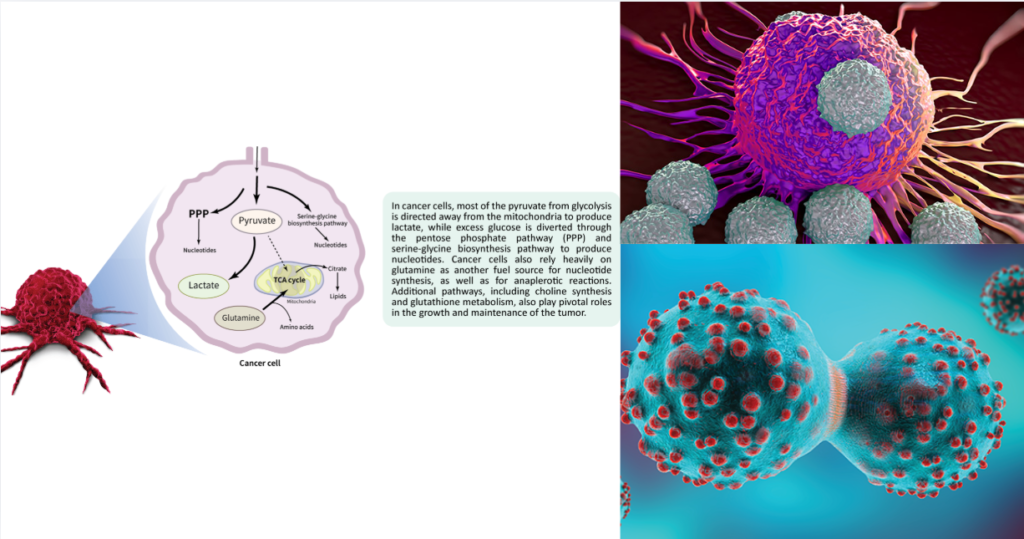
Cancer cells have a remarkable ability to rewire their metabolism to support rapid growth. Unlike healthy cells, which rely primarily on oxygen-dependent energy production, tumor cells often shift toward alternative energy pathways—even when oxygen is available.
The Warburg Effect
One hallmark of cancer metabolism is the Warburg effect, named after Otto Warburg, who first observed that cancer cells preferentially use glycolysis for energy rather than oxidative phosphorylation, even in oxygen-rich environments.
- Glycolysis: A process that breaks down glucose for quick energy.
- Advantage for tumors: Provides both energy and essential building blocks for rapid cell division.
Metabolic Plasticity
Tumors are metabolically flexible. Depending on nutrient availability and oxygen levels, they can switch between glycolysis, fatty acid oxidation, and amino acid metabolism. This adaptability is a major reason why cancers can survive in harsh conditions and resist certain treatments.
Key Fuels for Different Tumor Types
Different tumors rely on distinct energy sources based on their environment and genetic mutations.
Glucose-Dependent Tumors
Many fast-growing tumors, such as breast and colon cancers, heavily rely on glucose for energy.
- Why it matters: High glucose uptake can be detected using PET scans.
- Therapeutic angle: Drugs targeting glycolysis can slow growth in glucose-dependent tumors.
Lipid-Fueled Tumors
Some cancers, including prostate and ovarian cancers, can utilize fats as a primary energy source.
- Mechanism: Tumor cells break down fatty acids to generate ATP and metabolic intermediates.
- Implication: Limiting lipid availability or targeting fatty acid metabolism is a promising therapeutic approach.
Amino Acid Addiction
Certain tumors, like pancreatic cancer, depend on amino acids such as glutamine for growth.
- Glutamine use: Supports energy production and the synthesis of nucleotides and antioxidants.
- Potential treatment: Drugs targeting glutamine metabolism may selectively starve these tumors.
Hybrid Metabolism
Some aggressive cancers, including glioblastomas, can switch seamlessly between glucose, fats, and amino acids, making them particularly resilient to standard therapies.
How Tumor Microenvironment Influences Metabolism
The metabolic behavior of a tumor is not solely determined by its genetics; the tumor microenvironment also plays a critical role.
Oxygen Availability
- Hypoxia (low oxygen): Tumors in low-oxygen areas rely more on glycolysis.
- Normoxia (normal oxygen): Some tumors may still prefer glycolysis due to genetic mutations.
Nutrient Scarcity
Limited nutrient supply forces tumors to scavenge alternate fuels such as lactate, fatty acids, or extracellular proteins, demonstrating metabolic ingenuity.
Interactions With Surrounding Cells
- Cancer-associated fibroblasts and immune cells can release metabolites that tumors use for growth.
- These interactions can create a supportive ecosystem for cancer proliferation.
Clinical Implications of Cancer Metabolism
Understanding what fuels tumors opens doors for innovative diagnostic and treatment strategies.
Diagnostic Applications
- PET scans use radiolabeled glucose to detect tumors that preferentially consume glucose.
- Metabolic profiling helps predict tumor aggressiveness and potential treatment responses.
Targeted Therapies
- Glycolysis inhibitors: Drugs that block glucose breakdown.
- Fatty acid metabolism inhibitors: Limit energy supply in lipid-dependent tumors.
- Amino acid restriction therapies: Starve tumors addicted to glutamine or other key nutrients.
Personalized Medicine
Metabolic profiling allows oncologists to tailor treatment based on the tumor’s specific fuel preferences, potentially improving outcomes.
Overcoming Challenges in Targeting Cancer Metabolism
Tumor metabolism is highly adaptable, which presents challenges for therapy.
Metabolic Redundancy
- Tumors can switch between fuel sources if one pathway is blocked.
- Combination therapies targeting multiple metabolic routes are often necessary.
Side Effects
- Many metabolic pathways are also active in normal cells.
- Selectivity is crucial to minimize toxicity.
Evolving Resistance
- Tumors can evolve resistance by mutating key enzymes or activating alternative pathways.
- Ongoing research focuses on dynamic treatment strategies to anticipate these changes.
Maximizing Insights From Cancer Metabolism Research
Researchers continue to explore how metabolic differences shape tumor behavior.
Integrating Metabolism With Genetics
- Combining genomic profiling with metabolic analysis provides a fuller picture of tumor biology.
- Helps identify vulnerabilities for targeted therapy.
Lifestyle Considerations
- Emerging research examines how diet, exercise, and metabolic health influence tumor growth.
- While not a standalone therapy, metabolic interventions may support traditional treatments.
Future Directions
- Immunometabolism: Understanding how tumor metabolism affects immune cell function.
- Metabolic imaging: Advanced techniques for monitoring treatment response in real time.
- Synthetic lethality approaches: Exploiting unique metabolic dependencies of tumors.
Conclusion
Cancer metabolism is a dynamic, complex process that fuels tumor growth in unique ways. Some tumors rely on glucose, others on fats or amino acids, and many can switch between energy sources depending on their environment. By understanding these metabolic strategies, researchers and clinicians are developing targeted therapies that exploit tumor vulnerabilities.
For patients and caregivers, this knowledge emphasizes the importance of personalized medicine and metabolic profiling. With ongoing research, the future of cancer treatment increasingly focuses on disrupting the fuel lines that tumors need to survive—empowering better outcomes and innovative solutions.
Cancer may be formidable, but uncovering what fuels each tumor brings us one step closer to more effective, tailored interventions.
Important Disclaimer: This article is for informational purposes only and should not replace professional advice. For health-related topics, consult healthcare providers. Individual results may vary, and personal circumstances should always be considered when implementing any suggestions.